Export Packaging That Prevents Rejection , Damage & Audit Failure
Engineered ISPM-15 compliant packaging designed for pharma and chemical exports.
Destinations
Compliant
Standards
Packaging
Most Export Packaging Fails Before
the Container Leaves the Port
Most Export Packaging Fails Before the Container Leaves the Port
If you export pharmaceutical or chemical products, you’ve likely seen this:
- Packaging rejected during QA or customs inspections
- Compliance requirements interpreted differently by suppliers and inspectors
- Product damage caused by moisture, vibration, or insufficient load design
- Missing or incomplete documentation during audits
One weak point in packaging is enough to delay shipments, trigger penalties, or damage overseas customer relationships.
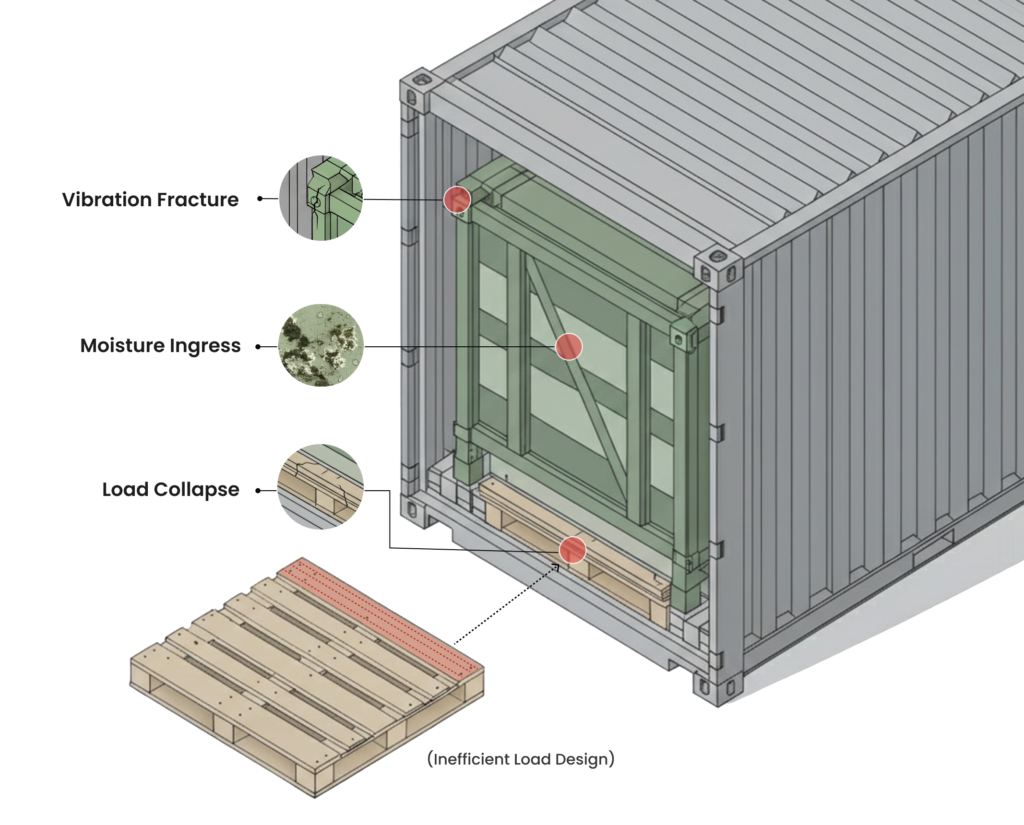
Load calculated for stacking and forklift handling

Designed for long sea transit and humidity exposure
The Problem Isn’t the Product.
It’s Commodity Packaging.
The Problem Isn’t the Product. It’s Commodity Packaging.
Most packaging suppliers sell standard pallets and crates. They optimize for price, not risk. For regulated industries, that approach fails. Pharma and chemical exports require engineered packaging, not generic wood solutions.
Packaging Engineered for Regulated Exports
Greenstrand designs export packaging the way engineers approach risk:
- Packaging designed around load, transport mode, and destination
- Material selection focused on moisture, contamination, and stability risks
- Crates and pallets calculated — not guessed
- Documentation prepared to support audits and inspections
The result: fewer discussions at customs and fewer surprises after dispatch.
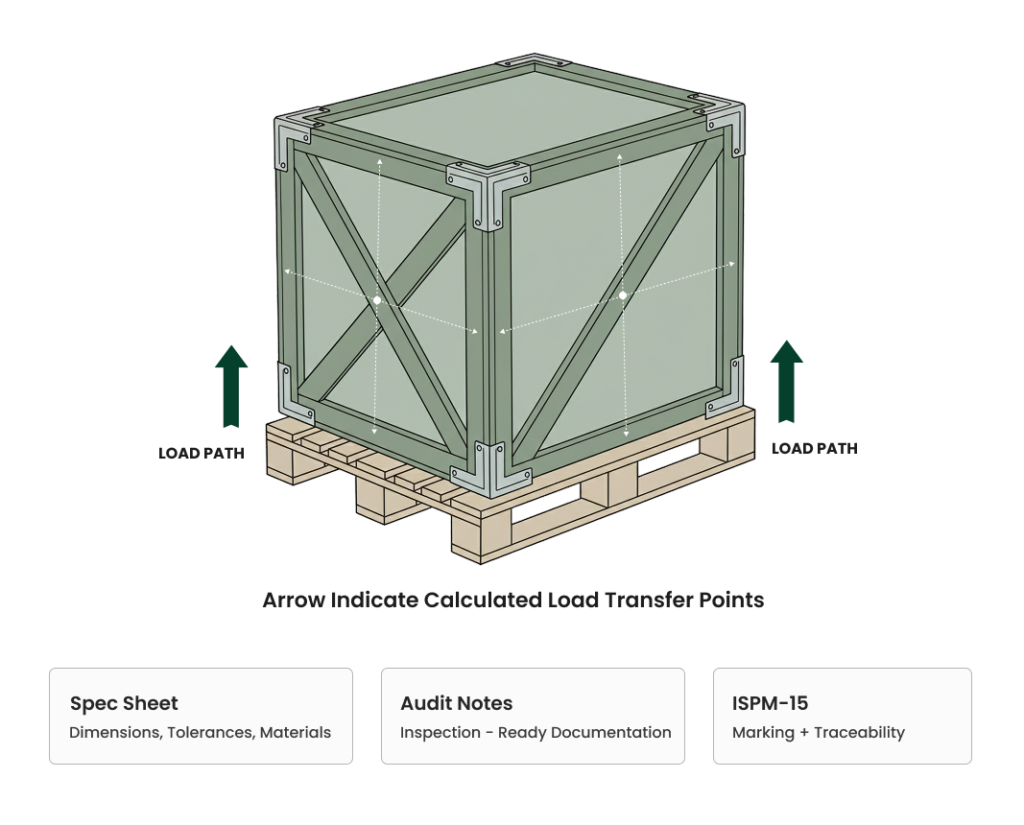
Protect sensitive cargo from moisture and handling damage
German Engineering Discipline. Indian Manufacturing Execution.
Greenstrand was founded by German engineers with experience in export-critical industries and international supply chains.
- Clear specifications instead of vague promises
- Calculations instead of assumptions
- Repeatable processes instead of improvisation

All wooden components are stamped and documented as per ISPM-15 requirements.
Manufacturing and execution in India, aligned to EU and US export expectations.
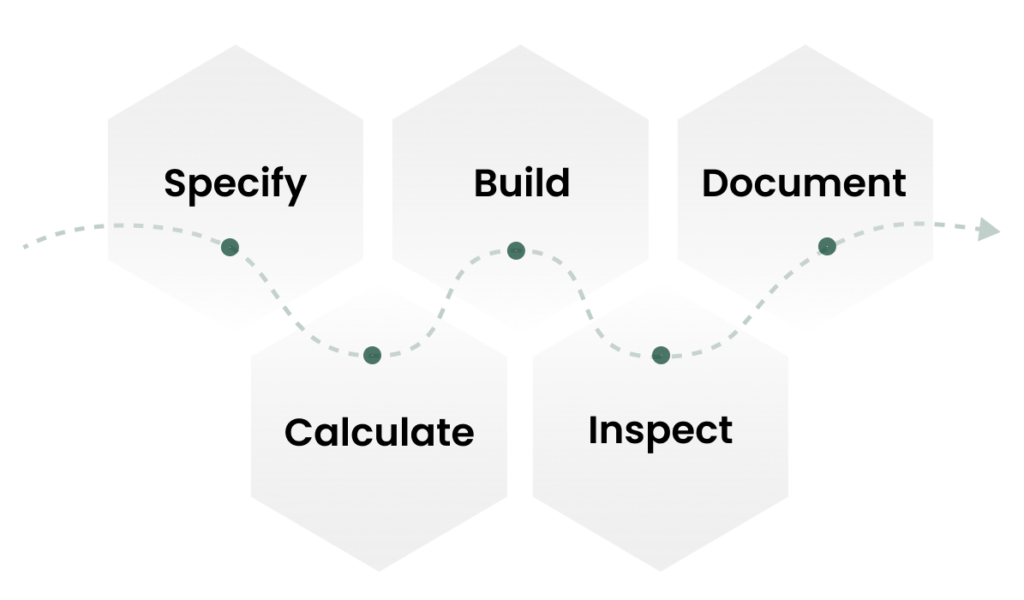
A repeatable approach
This isn’t about adding complexity. It’s about removing ambiguity.
Our Packaging Design Process
This isn't about adding complexity. It's about removing ambiguity at every stage of your export chain.
Who This Is For
This solution is designed for companies that:
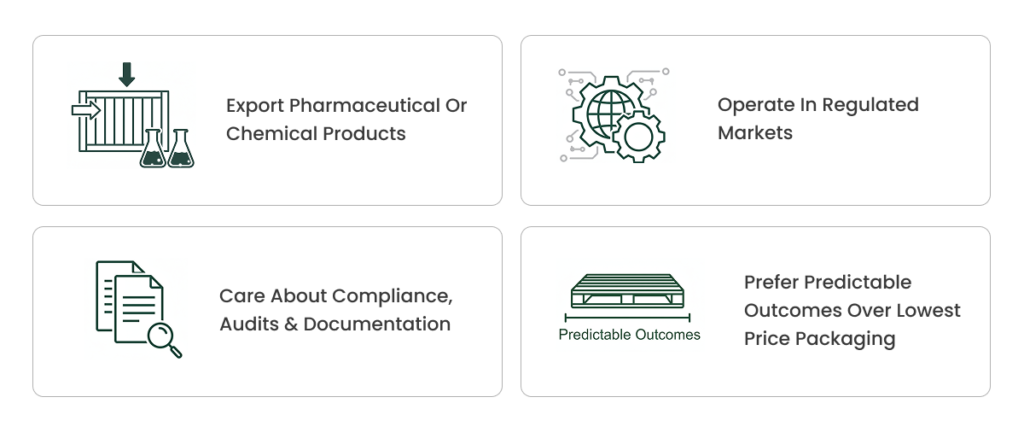
If you are only looking for the cheapest pallet, this is probably not a fit.
FREE CHECKLIST
Pharma & Chemical Export Packaging Readiness
Before your next shipment, verify:
- Are your pallets and crates compliance-ready?
- Will your packaging pass overseas QA checks?
- Is your documentation audit-safe?
No sales pitch. Just the requirements overseas customers and inspectors actually check.

Get In Touch
Tell Us About Your Shipment
Share your packaging requirements — cargo type, destination, dimensions, compliance needs. We’ll review and come back with a specific recommendation for your export route.
- +91 8448581799 · 01 244251686
- sales@greenstrand.in
- Unit 1003 A&B, JMD Regent Square, DLF Phase 2, Gurugram, Haryana 122002
